Well, I just saw that researchers had succeeded in photolysis of water by solar energy thanks to a semi-conductor: titanium dioxide.
Basically the water passes through a glass tube in which there is the semiconductor and the sun does the rest.
In norway do they practice photovoltaic electrolysis, and what if we did it here too?
I think I dream, we have a hand of inexhaustible energy and we continue to spend billions for nuclear energy?
Between ITER which is likely to be a fiasco and the EPR which has an invoice has lengthened without speaking about the old power stations which will listen to a max to dismantle (if one does it one day), or to decontaminate (if one does not do it) ; in this second case the bill may cost a lot in lives too!
And here they go to look for oil in Guyana now, and still billions of dollars that the taxpayer will pay, not to mention the non-economic fallout on Guyana precisely: since when do people benefit from the resources of their own ground ?
Do not dream either!
Photolysis of hydrogen with titanium dioxide
There are plenty in the paintings, the sunscreens of this TiO2, and there are plenty of studies like this, but their use remains in the publications.
http://phys.org/news10244.html
http://phys.org/news122534699.html
http://phys.org/news135349137.html
http://phys.org/news138873713.html
etc ..
but between the first possibilities and the industrial use, there is a world full of failures, considering the complexity of the photochemical reactions.
http://phys.org/news10244.html
http://phys.org/news122534699.html
http://phys.org/news135349137.html
http://phys.org/news138873713.html
etc ..
but between the first possibilities and the industrial use, there is a world full of failures, considering the complexity of the photochemical reactions.
0 x
- plasmanu
- Econologue expert

- posts: 2847
- Registration: 21/11/04, 06:05
- Location: The 07170 Lavilledieu viaduct
- x 180
interested 
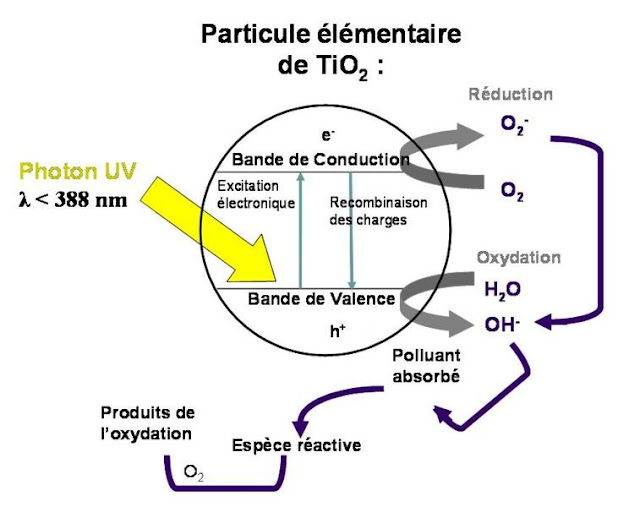
TiO2 in powder form, in a liquid medium will oxidize large molecules in contact with UV.
It is mainly used to oxidize chemicals.
It's photocatalysis.
http://fr.ekopedia.org/Photocatalyse
A beautiful concrete example:
http://photocal.over-blog.com/pages/La_ ... 40327.html
Photodecomposition study with TiO2 in aqueous solution: 13 page
https://www.econologie.info/share/partag ... FKF7AG.pdf
Titanium dioxide
http://reade.com/fr/products/35-oxides- ... D1&page%3D
TiO2 in powder form, in a liquid medium will oxidize large molecules in contact with UV.
It is mainly used to oxidize chemicals.
It's photocatalysis.
http://fr.ekopedia.org/Photocatalyse
A beautiful concrete example:
http://photocal.over-blog.com/pages/La_ ... 40327.html
Photodecomposition study with TiO2 in aqueous solution: 13 page
https://www.econologie.info/share/partag ... FKF7AG.pdf
Titanium dioxide
http://reade.com/fr/products/35-oxides- ... D1&page%3D
0 x
"Not to see Evil, not to hear Evil, not to speak Evil" 3 little monkeys Mizaru
- plasmanu
- Econologue expert

- posts: 2847
- Registration: 21/11/04, 06:05
- Location: The 07170 Lavilledieu viaduct
- x 180
The problem is that titanium dioxide will only provide oxygen.
TiO2 is the anode.
Hydrogen is formed at the cathode.
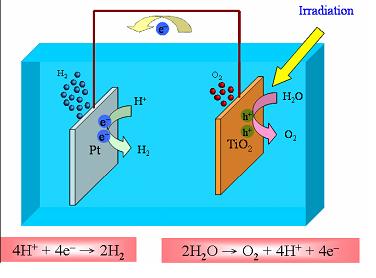
http://dioxydedetitanetpe.blogspot.fr/2 ... ie-ii.html
TiO2 is the anode.
Hydrogen is formed at the cathode.

http://dioxydedetitanetpe.blogspot.fr/2 ... ie-ii.html

0 x
"Not to see Evil, not to hear Evil, not to speak Evil" 3 little monkeys Mizaru
Plasmanu has gone into the theory and the well-known bases of TiO2, with dyes that fade in the sun all by themselves, even without TiO2, spontaneously less quickly.
But Plasmanu did not read the scientific links with the publications, which I put, (Vol.6 No. 2 issue of Nano Letters, the Grimes' team, which includes Gopal K. Mor, Karthik Shankar, Maggie Paulose, and Oomman K. Varghese), much more complex, nanometric to use separate loads, such as photosynthesis mechanisms, algae and plants, very real and not theoretical, that we seek to copy with better performance.
But Plasmanu did not read the scientific links with the publications, which I put, (Vol.6 No. 2 issue of Nano Letters, the Grimes' team, which includes Gopal K. Mor, Karthik Shankar, Maggie Paulose, and Oomman K. Varghese), much more complex, nanometric to use separate loads, such as photosynthesis mechanisms, algae and plants, very real and not theoretical, that we seek to copy with better performance.
The biggest hurdle to overcome the photocatalytic splitting of water has been the lack of a robust catalyst that oxidizes water. In fact, the best known catalyst, which is very effective when irradiated with visible light, is a manganese-containing enzyme in the photosynthetic apparatus of living organisms.
0 x
- plasmanu
- Econologue expert

- posts: 2847
- Registration: 21/11/04, 06:05
- Location: The 07170 Lavilledieu viaduct
- x 180
dedeleco wrote: and there are plenty of studies of this kind, but their use remains in the publications.
And that's concrete:
without producing H2
23w for 200m²
http://fr.aliexpress.com/product-fm/479 ... alers.html


0 x
"Not to see Evil, not to hear Evil, not to speak Evil" 3 little monkeys Mizaru
- chatelot16
- Econologue expert

- posts: 6960
- Registration: 11/11/07, 17:33
- Location: Angouleme
- x 264
it smells like very expensive stuff that produces very little
at home I have a solar collector that instead of producing hydrogen difficult to store, produce usable carbohydrate fuel easy to store ... and even sometimes edible!
these sensors do not have a huge yield but an unbeatable value for money: they grow by themselves!
at home I have a solar collector that instead of producing hydrogen difficult to store, produce usable carbohydrate fuel easy to store ... and even sometimes edible!
these sensors do not have a huge yield but an unbeatable value for money: they grow by themselves!
0 x
Back to "biofuels, biofuels, biofuels, BtL, non-fossil alternative fuels ..."
Who is online ?
Users browsing this forum : No registered users and 142 guests

